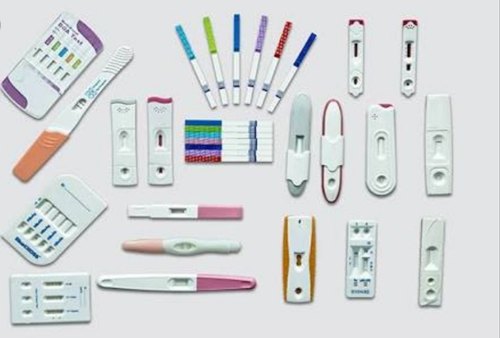
COLOMBO – Sri Lanka is set to use rapid antigen testing to detect COVID-19 patients as the number of people infected with the virus continues to increase exponentially.
Dr. Ananda Wijewickrema of the National Medicines Regulatory Authority (NMRA), in an interview on a private television channel on Monday (26), said health authorities had on Friday (23) given its approval to order the new rapid antigen testing kits. The World Health Organization (WHO) has already approved the use of rapid antigen testing as a means of confirming COVID-19 positive cases.
According to Dr. Wijewickrema, Sri Lanka is expected to receive 20,000 test kits as the first batch from the WHO in the coming days . He also said the State Pharmaceuticals Corporation has been tasked with ordering them.
Dr. Wijewickrema said the NMRA had previously rejected many antigen tests for COVID-19 proposed by various parties, as they had low accuracy. However, as the rapid antigen testing in question is already WHO approved the NMRA quickly approved it, he added.
He said the cost of rapid antigen test kits were very low and test results can be obtained within 20 minutes, adding that using rapid antigen testing was a good solution to the increase of patients, as results can be obtained almost immediately.
Dr. Wijewickrema explained the test kits were expected to be used to test contacts of already identified COVID patients, to test people showing up at hospital OPDs with symptoms similar to COVID, and to conduct random tests in the community.
According to WHO, the rapid antigen tests target virus proteins and somewhat resemble pregnancy tests.
The test kits are not yet available at pharmacies and can only be administered by trained medical personnel. With a nasal or throat swab, testers can find out within approximately 15 minutes whether a patient has contracted the SARS-CoV-2 virus and is infectious and must be quarantined.
The WHO also announced that it will provide 120 million rapid antigen tests to people in lower- and middle-income countries over the next six months. Test kits manufacturers, South Korea’s SD BioSensor and US-based Abbott Laboratories have also agreed to provide 20% of their test kits to lower- and middle-income countries.
-economynext.com